Research
Activation of the ER stress sensor IRE1α by metastable and unfolded proteins
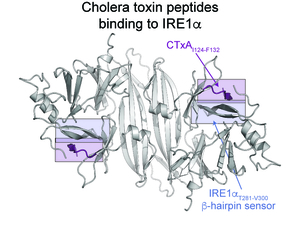
The goal of this project is to understand how the ER stress sensor IRE1α detects features of metastable and unfolded proteins as signs of ER stress to activate an unfolded protein response (UPR) and affect protein folding and quality control. We will integrate biophysical, biochemical, and cell biological approaches to (1) define what structural features of ER luminal proteins IRE1α recognizes as signs of ER stress; (2) determine how IRE1α recognizes unfolded proteins to initiate the UPR; and (3) test if IRE1α can act as a chaperone to prevent aggregation of unfolded proteins and affect quality control. Results from these studies will uncover new mechanisms for tools and therapeutics to modulate IRE1α function in cellular models and human disease.
Function and evolution of IRE1β in epithelial ER stress response and mucosal homeostasis
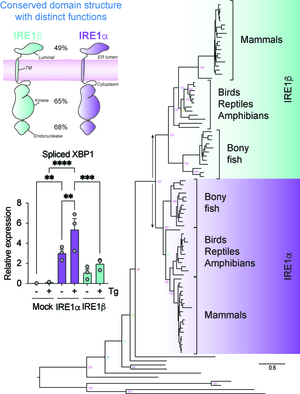
The goal of this project is to understand how the epithelial-specific ER stress sensor IRE1β operates in ER proteostasis and in MUC2 biosynthesis and secretion in intestinal epithelial cells. We will (1) define the structural features that explain the divergent enzymatic activities of IRE1β and how they affect the ER stress response in epithelial cells; (2) identify the features of folded and unfolded proteins that IRE1β recognizes in intestinal epithelial cells as signs of ER stress; and (3) determine how IRE1β regulates the folding and secretion of MUC2 in epithelial cells. Dysregulated UPR signaling and unresolved ER stress are hallmarks of inflammatory diseases, including inflammatory bowel disease. Understanding what IRE1β does and how IRE1β affects ER proteostasis in intestinal epithelial cells will provide mechanistic insight for how epithelial cells have adapted to accommodate exposure to environmental factors, and how dysfunction contributes to disease pathophysiology.
IRE1β enables host-microbiota crosstalk to protect against colitis
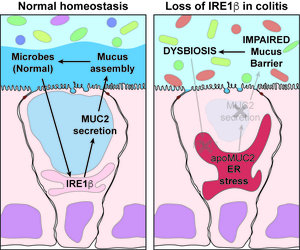
The goal of this project is to test if a defective host-microbiota feedback loop mediated by the ER stress sensor IRE1β underlies the goblet cell dysfunction, abnormal mucus barrier, and the resulting microbial dysbiosis that contribute to the pathogenesis of ulcerative colitis (UC). Using human samples, we will test if loss of IRE1β expression causes accumulation of MUC2 and ER stress in goblet cells in UC and if dysbiotic microbial communities fail to induce IRE1β expression and mucus barrier function using in vitro and in vivo models. These studies will validate an IRE1β-dependent microbe-goblet cell-mucus feedback loop in maintaining epithelial barrier function and host defense in the human colon and contributing to the pathophysiology of UC when dysfunctional. This is a necessary step for clinical translation and will enable follow-on studies to identify small molecules and microbial factors that can be used therapeutically to target this pathway in UC.
Recent Publications
- IRE1α recognizes a structural motif in cholera toxin to activate an unfolded protein response.
- Mechanisms underlying distinct subcellular localization and regulation of epithelial long myosin light-chain kinase splice variants.
- p120 RasGAP and ZO-2 are essential for Hippo signaling and tumor-suppressor function mediated by p190A RhoGAP.
