Thermoregulation
Related Publications
-
Ramirez-Plascencia, O. D., De Luca, R., Machado, N. L., Eghlidi, D., Khanday, M. A., Bandaru, S. S., Raffin, F., Vujovic, N., Arrigoni, E., & Saper, C. B. (Submitted). A hypothalamic circuit for circadian regulation of corticosterone secretion. Research Square, rs.3.rs-4718850.
Abstract
Last updated on 06/04/2025The secretion of cortisol in humans and corticosterone (Cort) in rodents follows a daily rhythm which is important in readying the individual for the daily active cycle and is impaired in chronic depression. This rhythm is orchestrated by the suprachiasmatic nucleus (SCN) which governs the activity of neurons in the paraventricular nucleus of the hypothalamus that produce the corticotropin-releasing hormone (PVHCRH neurons). The dorsomedial nucleus of the hypothalamus (DMH) serves as a crucial intermediary, being innervated by the SCN both directly and via relays in the subparaventricular zone, and projecting axons to the PVH, thereby exerting influence over the cortisol/corticosterone rhythm. However, the role and synaptic mechanisms by which DMH neurons regulate the daily rhythm of Cort secretion has not been explored. We found that either ablating or acutely inhibiting the DMH glutamatergic (DMHVglut2) neurons resulted in a 40-70% reduction in the daily peak of Cort. Deletion of the Vglut2 gene within the DMH produced a similar effect, highlighting the indispensable role of glutamatergic signaling. Chemogenetic stimulation of DMHVglut2 neurons led to an increase of Cort levels, and optogenetic activation of their terminals in the PVH in hypothalamic slices directly activated PVHCRH neurons through glutamate release (the DMHVglut2 → PVHCRH pathway). Similarly, ablating, inhibiting, or disrupting GABA transmission by DMH GABAergic (DMHVgat) neurons diminished the circadian peak of Cort, particularly under constant darkness conditions. Chemogenetic stimulation of DMHVgat neurons increased Cort, although with a lower magnitude compared to DMHVglut2 neuron stimulation, suggesting a role in disinhibiting PVHCRH neurons. Supporting this hypothesis, we found that rostral DMHVgat neurons project directly to GABAergic neurons in the caudal ventral part of the PVH and adjacent peri-PVH area (cvPVH), which directly inhibit PVHCRH neurons, and that activating the DMHVgat terminals in the cvPVH in brain slices reduced GABAergic afferent input onto the PVHCRH neurons. Finally, ablation of cvPVHVgat neurons resulted in increased Cort release at the onset of the active phase, affirming the pivotal role of the DMHVgat → cvPVHVgat → PVHCRH pathway in Cort secretion. In summary, our study delineates two parallel pathways transmitting temporal information to PVHCRH neurons, collectively orchestrating the daily surge in Cort in anticipation of the active phase. These findings are crucial to understand the neural circuits regulating Cort secretion, shedding light on the mechanisms governing this physiological process and the coordinated interplay between SCN, DMH, and PVH.
-
Machado, N. L., Lynch, N., Costa, L. H. A., Melville, D., Kucukdereli, H., Kaur, S., Banks, A. S., Raffin, F., Ramirez-Plascencia, O. D., Aten, S., Lima, J. D., Bandaru, S. S., Palmiter, R. D., & Saper, C. B. (2025). Preoptic EP3R neurons constitute a two-way switch for fever and torpor. Nature.
Abstract
Last updated on 06/04/2025"Many species use a temporary decrease in body temperature and metabolic rate (torpor) as a strategy to survive food scarcity in a cool environment. Torpor is caused by preoptic neurons that express a variety of peptides and receptors1–7 , but no single genetic marker has been found for this population. Here we report that expression of the prostaglandin EP3 receptor (EP3R) marks a unique population of median preoptic nucleus (MnPO) neurons that are required for both torpor and lipopolysaccharideinduced fever8 . The MnPO-EP3R neurons produce persistent fever responses when inhibited and prolonged hypothermic responses when activated either chemogenetically or optogenetically, even for brief periods of time. The mechanism for these prolonged responses appears to involve increases in intracellular levels of cAMP and calcium that may persist for many minutes up to hours beyond the termination of a stimulus. These properties endow the population of MnPO-EP3R neurons with the ability to act as a two-way switch for the hypothermic and hyperthermic responses that are required for survival."
-
Heyward, F. D., Liu, N., Jacobs, C. L., Machado, N. L., Ivison, R., Uner, A., Srinivasan, H., Patel, S. J., Gulko, A., Sermersheim, T. J., Tsai, L., & Rosen, E. (2024). AgRP neuron cis-regulatory analysis across hunger states reveals that IRF3 mediates leptin’s acute effects. Nature Communication, 15(1), 4646.
Abstract
Last updated on 06/04/2025AgRP neurons in the arcuate nucleus of the hypothalamus (ARC) coordinate homeostatic changes in appetite associated with fluctuations in food availability and leptin signaling. Identifying the relevant transcriptional regulatory pathways in these neurons has been a priority, yet such attempts have been stymied due to their low abundance and the rich cellular diversity of the ARC. Here we generated AgRP neuron-specific transcriptomic and chromatin accessibility profiles from male mice during three distinct hunger states of satiety, fasting-induced hunger, and leptin-induced hunger suppression. Cis-regulatory analysis of these integrated datasets enabled the identification of 18 putative hunger-promoting and 29 putative hunger-suppressing transcriptional regulators in AgRP neurons, 16 of which were predicted to be transcriptional effectors of leptin. Within our dataset, Interferon regulatory factor 3 (IRF3) emerged as a leading candidate mediator of leptin-induced hunger-suppression. Measures of IRF3 activation in vitro and in vivo reveal an increase in IRF3 nuclear occupancy following leptin administration. Finally, gain- and loss-of-function experiments in vivo confirm the role of IRF3 in mediating the acute satiety-evoking effects of leptin in AgRP neurons. Thus, our findings identify IRF3 as a key mediator of the acute hunger-suppressing effects of leptin in AgRP neurons.
-
Last updated on 06/04/2025Saper, C. B., & Machado, N. L. (2022). Identifying specific populations of preoptic thermoregulatory neurons: The way forward. Temperature (Austin), 9(1), 12-13.
-
Machado, N. L., & Saper, C. B. (2022). Genetic identification of preoptic neurons that regulate body temperature in mice. Temperature (Austin), 9(1), 14-22.
Abstract
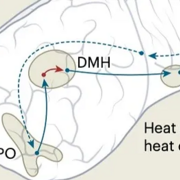
Last updated on 06/04/2025There has been an explosion recently in our understanding of the neuronal populations in the preoptic area involved in thermoregulation of mice. Recent studies have identified several genetically specified populations of neurons predominantly in the median preoptic nucleus (MnPO) but spreading caudolaterally into the preoptic area that regulate body temperature. These include warm-responsive neurons that express the peptides PACAP, BDNF, or QRFP; and receptors for temperature, leptin, estrogen, or prostaglandin E2 (PGE2). These neurons are predominantly glutamatergic and driving them opto- or chemogenetically can cause profound hypothermia, and in some cases, periods of torpor or a hibernation-like state. Conversely, fever response is likely to depend upon inhibiting the activity of these neurons through the PGE2 receptor EP3. Another cell group, the Brs3-expressing MnPO neurons, are apparently cold-responsive and cause increases in body temperature. MnPOQRFP neurons cause hypothermia via activation of their terminals in the region of the dorsomedial nucleus of the hypothalamus (DMH). As the MnPOQRFP neurons are essentially glutamatergic, and the DMH largely uses glutamatergic projections to the raphe pallidus to increase body temperature, this model suggests the existence of local inhibitory interneurons in the DMH region between the MnPOQRFP glutamatergic neurons that cause hypothermia and the DMH glutamatergic neurons that cause hyperthermia. The new genetically targeted studies in mice provide a way to identify the precise neuronal circuitry that is responsible for our physiological observations in this species, and will suggest critical experiments that can be undertaken to compare these with the thermoregulatory circuitry in other species.
-
Saper, C. B., & Machado, N. L. (2020). Flipping the switch on the body’s thermoregulatory system. Nature, 583(7814), 34-35.
Abstract
A population of excitatory neurons has been found to have a key role in controlling body temperature in rodents. The discovery adds to a body of work that is raising questions about long-standing models of thermoregulation.
Last updated on 06/04/2025