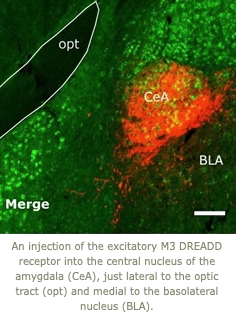
 1. Cataplexy is generally triggered by strong, positive emotions such as those that occur when laughing at a great joke or playing with a friend. The amygdala is a key site through which emotions trigger motor responses, and we are working to identify just which neurons in the amygdala mediate cataplexy. Using engineered DREADD receptors, we found that activation of GABAergic neurons in the central nucleus of the amygdala roughly doubles the amount of cataplexy whereas inhibition of these cells reduces cataplexy. We are now finding that cataplexy is driven by a subset of these neurons that are activated by oxytocin. In people with narcolepsy, cataplexy almost always occurs during social interaction, and these findings suggest that higher oxytocin tone when socializing may increase the likelihood of cataplexy.
1. Cataplexy is generally triggered by strong, positive emotions such as those that occur when laughing at a great joke or playing with a friend. The amygdala is a key site through which emotions trigger motor responses, and we are working to identify just which neurons in the amygdala mediate cataplexy. Using engineered DREADD receptors, we found that activation of GABAergic neurons in the central nucleus of the amygdala roughly doubles the amount of cataplexy whereas inhibition of these cells reduces cataplexy. We are now finding that cataplexy is driven by a subset of these neurons that are activated by oxytocin. In people with narcolepsy, cataplexy almost always occurs during social interaction, and these findings suggest that higher oxytocin tone when socializing may increase the likelihood of cataplexy.
2. While the orexin neuropeptides are best understood in promoting wakefulness and regulating REM sleep, they also have many other behavioral effects, including effects on breathing. We are now studying how loss of orexin signaling affects breathing, and how restoration of orexin signaling with orexin agonists improves breathing. Orexins likely excite many brainstem regions that influence breathing, and by focally expressing orexin receptors in certain brainstem regions, we are also working to define the specific circuits through which this occurs.
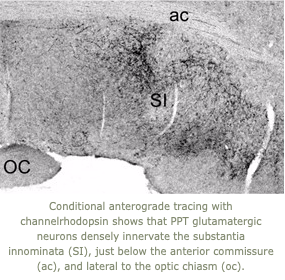
3. Many people with narcolepsy have difficulty maintaining attention for long periods which impacts their performance in school and work and when driving. The orexins excite neurons in the cortex, basal forebrain, and many other brain regions which promote normal cognition. Using GRAB (GPCR activation-based) sensors and fiber photometry, we are measuring acetylcholine tone across a variety of conditions, and how this is impacted by orexin signaling with the goal of better defining how orexin deficiency impairs cognition.
4. With our colleague Carrie Mahoney, we are studying the neural mechanisms through which menopause disrupts sleep. Removing the ovaries from female mice triggers hot flashes and fragments sleep, and we are examining whether hypothalamic circuits including the the KNDy neurons mediate these troublesome symptoms.
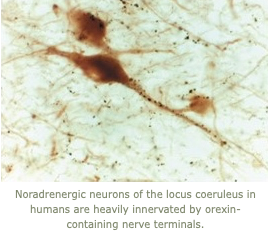
5. In collaboration with our colleagues Elda Arrigoni and Vetri Ramalingam, we are studying the neurobiological mechanisms of current and future medications for treating narcolepsy. Specifically, we are using GRAB sensors to determine if pitolisant (a drug that increases histamine and other monoamines) promotes wakefulness by inhibiting brain sleep mechanisms, and we are investigating the effects of a medication that enhances signaling through melanin-concentrating hormone receptors.
6. Past projects have included studies of human brains and the interactions of sleep and pain. In addition, we found that loss of the orexin neurons in people with narcolepsy is also accompanied by a large increase in the number of neurons producing histamine. This may be a compensatory response that helps produce wakefulness after loss of the orexin neurons. In mouse experiments, we found that reductions in sleep increase behavioral responses to painful stimuli, and these responses are normalized with wake-promoting medications. In addition, we found that mice with neuropathic pain have fragmented sleep, which may be a useful biomarker of spontaneous pain.
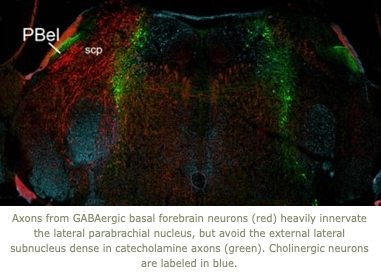
Our lab uses a variety of anatomic, physiologic, and molecular techniques. We frequently study sleep/wake behavior in mice using detailed analysis of the electroencephalogram in conjunction with optogenetics, photometry with GCaMP and GRAB sensors, chemoactivation studies using DREADDs (designer receptor exclusively activated by designer drugs), recordings of muscle activity, locomotion, behavior, and body temperature. We have also developed new mathematical techniques for analysis of the transitions between behavioral states and examination of intermediate states. We trace neural pathways using novel and conventional anterograde and retrograde tracers, and we perform immunostaining and in situ hybridization histochemistry to map the distribution of neurotransmitters, receptors, and other molecules. We also use a variety of molecular techniques to design and produce novel recombinant mice.
Through these approaches, we hope to gain a detailed understanding of orexin neurobiology that will result in highly effective therapies for patients with narcolepsy and more broadly, enhance our knowledge of sleep.
