Stroke is the 4th leading cause of death and a leading cause of disability in the United States (US). There are approximately 700,000 new ischemic strokes per year and about 7 million people living with chronic ischemic stroke disability in the US. The vast majority (70-85%) of first strokes result in hemiplegia and/or hemiparesis and at 6 months after stroke only 60% of stroke sufferers have achieved independence in their activities of daily living. In the US, annual stroke costs exceed $100 billion with the majority stemming from chronic stroke care.
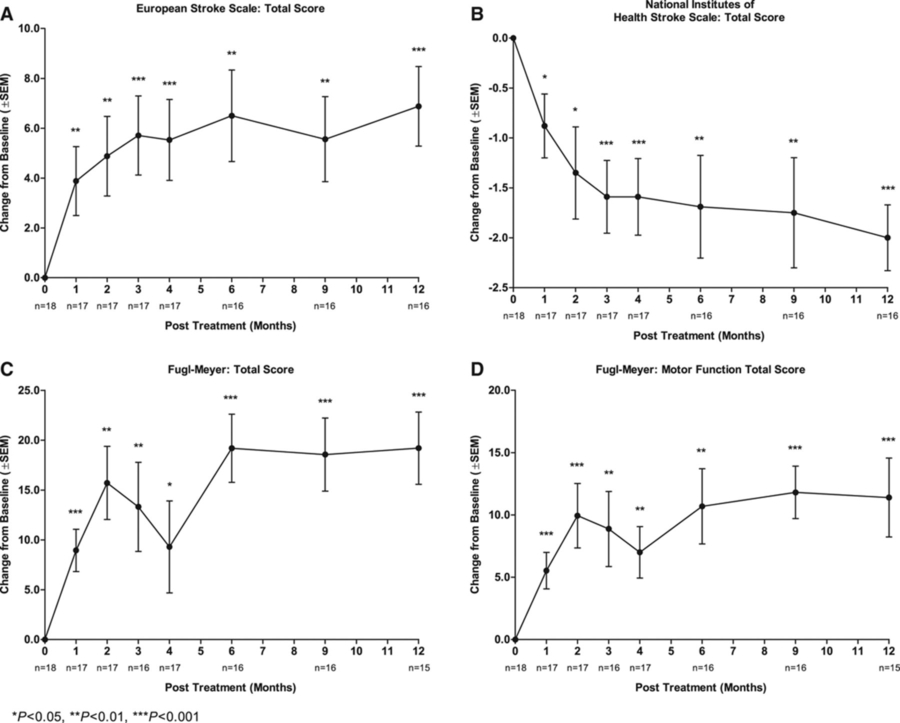
NR1 neural stem cells were developed to stimulate and promote the brain's native repair and regenerative medicines. Developed by Gary Steinberg, chair emeritus at Stanford and collaborators, NR1 is specifically targeted to the subcortical brain tissue adjacent to the subcortical region of the index stroke. This area is active in post-stroke tissue repair and brain reorganization in preclinical studies and on human functional brain imaging. The putative NR1 mechanism of action is not to replace and become new brain cells, but rather to secrete protein factors required to stimulate and nurture the brain’s native repair and regeneration mechanisms, including axonal/dendritic sprouting, synaptogenesis, angiogenesis, and modulation of inflammation. The stereotactic intracerebral NR1 administration in this study NR1-03 is consistent with preclinical pharmacology studies during which injection coordinates were identified that directly deliver NR1 into the tissue adjacent to subcortical and cortical stroke lesions. The current Phase 2b clinical trial follows the recent Phase 1/2a clinical study that demonstrated that NR1, at a dose comparable to that being studied in NR1-03, is generally safe and well-tolerated in adults with stable hemiparesis from chronic ischemic subcortical and/or cortical stroke. In the single-arm, dose-ranging Phase 1/2a study, NR1 demonstrated encouraging activity and restoration of lost motor function compared to baseline, pre-treatment exams, in the majority of evaluable subjects.
In this Phase 2b study, NR1-03, named suNR1se II, we will be enrolling patients to evaluate improvement in the total Fugl-Meyer Motor Scale (FMMS) induced by NR1 stereotactic intracerebral transplantation compared to control in adults with chronic ischemic subcortical and/or cortical stroke.
See this paper from 2016 for reference.